My Baby Has Lower Left Kidney Than Right
Abstract
Information about growth of kidney structures in early on life is express. In a population-based prospective cohort study, from foetal life onwards, we constructed reference curves for kidney growth from the third trimester of pregnancy until early on childhood, using data from 1,158 salubrious children. Kidney size, defined equally length, width, depth and book, was measured in the third trimester of pregnancy and at the postnatal ages of 6 months and 24 months. Analyses were based on more than ii,500 kidney measurements. In the third trimester of pregnancy and at half dozen months of age all kidney measurements were larger in boys than in girls. At 24 months of age, these gender differences were only significant for left kidney structures and right kidney length. Both groups showed trends towards smaller left kidney measurements than right kidney measurements at all ages. Gender-specific reference curves based on post-conceptional and postnatal ages were synthetic for left and right kidney length, width, depth and volume. We ended that kidney size is influenced by age and gender. Left kidney size tended to exist smaller than right kidney size, except for kidney length. The reference curves tin can exist used for assessing kidney structures by ultrasound in foetal life and early childhood.
Introduction
Assessment of kidney size in children is important for clinical and epidemiological studies. Aberrant early kidney development may have perinatal and neonatal consequences [1]. Smaller foetal kidney size has likewise been suggested to be related to hypertension and renal affliction in adulthood [2, 3]. Recently, we showed in the aforementioned accomplice that small kidney size in foetal life tends to persist in early babyhood. Furthermore, maternal anthropometric characteristics, foetal biometric data and blood period patterns were associated with kidney size in childhood. College growth rates in early childhood were positively associated with combined kidney volume [4]. These results suggest that variations in foetal and early on postnatal exposure and growth might have persistent consequences for kidney size. Kidney size can be measured non-invasively and efficiently with ultrasound. Few studies have published reference ranges for kidney size in healthy children during foetal and neonatal life [v, 6]. One study showed reference data on postnatal kidney growth from birth to xviii months of age [7]. Previous studies were based on the characteristics of postnatal kidney growth and focused more often than not on kidney book in relation to weight, height or body area. Recently, new reference centiles were generated to assess kidney size of children with 'single kidneys' to identify those patients with unfavourable courses and relevant single kidney growth impairment [eight]. Currently, there are no studies that have evaluated normal kidney growth from belatedly foetal life to early on babyhood. This perinatal period may be of importance in the identification of aberrant kidney size and growth, with subsequent short-term and long-term clinical consequences [9, x].
Therefore, the aim of this study was to construct reference curves for kidney structures including kidney length, width, depth and volume in children from the third trimester of pregnancy until the postnatal age of 24 months in a population-based cohort.
Methods
Study blueprint
This report was embedded in the Generation R Report, a population-based prospective cohort study from foetal life until young adulthood in Rotterdam, Kingdom of the netherlands [11, 12]. Detailed assessments of foetal and postnatal growth and development were conducted in a subgroup of one,232 mothers and their children [11, 12]. In this subgroup foetal kidney ultrasounds were performed in the 3rd trimester of pregnancy (gestational historic period 30 weeks) and postnatal kidney ultrasounds were performed at the ages of 6 months and 24 months. The study was approved by the Medical Ethics Committee of the Erasmus MC, Rotterdam. Written informed consent was obtained from all participants.
Population for analysis
In total, 1,232 women were enrolled in the Focus cohort. The analysis was limited to singleton live-born infants (n = 1,215) whose mothers had participated in the third trimester measurements. Twin pregnancies (north = fifteen) and pregnancies leading to perinatal decease (n = 2) were excluded from the analysis. Kidney ultrasounds were successfully performed in 95% (n = ane,158) of these subjects. Of the initial i,215 singleton alive-born infants, 74% (n = 901) and 70% (n = 856) underwent postnatal cess at the ages of vi months and 24 months, respectively. Kidney ultrasounds were successfully performed in 83% (north = 747) and fourscore% (n = 683) of these infants, respectively. Missing values were mainly due to the infants crying or to the unavailability of equipment or radiographer. Infants who had undergone a postnatal kidney ultrasound at the ages of 6 months and 24 months did not differ from the postnatal non-responders in foetal and birth characteristics. There were no kidney or ureterovesical anomalies other then mild pyelectasis over x mm (n = 3) in our study population. In total, analyses were based on more than ii,500 kidney measurements. The numbers of kidney growth measurements available for the analyses are shown in Table 1.
Ultrasound measurements
Gestational age was established past foetal ultrasound. Crown–rump length was used for pregnancy dating until a gestational age of 12 weeks and 5 days (crown–rump length smaller than 65 mm), and biparietal bore was used for pregnancy dating thereafter (gestational age from 12 weeks and 5 days onwards, biparietal diameter larger than 23 mm) [thirteen].
Foetal left and right kidneys were measured in the third trimester of pregnancy with an ATL-Philips HDI 5000 instrument (Seattle, WA, Us) equipped with a two.0–5.0 MHz curved array transducer. In a sagittal plane the maximum longitudinal kidney length was measured, with the calipers placed on the outer edges of the caudal and cranial sides. Antero-posterior (kidney width) and transverse (kidney depth) diameters were measured perpendicular to each other, outer to outer, in an axial plane [fourteen]. Values of maximum bipolar kidney length, width and depth were obtained from both the left and right kidneys. Kidney width and depth were measured at the level of the kidney hilum [14, 15]. The images were magnified to ensure optimal measurements [fourteen]. Foetal growth characteristics (head circumference, abdomen circumference, femur length) were measures at the same visit, and foetal weight was estimated [13].
Postnatally, two-dimensional ultrasounds of the kidneys were performed in children at the ages of 6 months and 24 months. The examination was carried out in a quiet room with the child quietly awake in a supine position. This position was standardised to forestall differences resulting from position [14, 15]. Mean length, width and depth were calculated as the boilerplate of three measurements and used for data analysis. Foetal and postnatal kidney volumes were both calculated in cubic centimetres using the equation of an ellipsoid: \( {\text{volume}}\;\left( {{\text{c}}{{\text{chiliad}}^3}} \correct) = 0.523 \times {\text{mean}}\;{\text{length}}\;\left( {\text{cm}} \correct) \times {\text{mean}}\;{\text{width}}\;\left( {\text{cm}} \right) \times {\text{mean}}\;{\text{depth}}\;\left( {\text{cm}} \right) \) [15, 16]. The infants' anthropometric parameters, including weight and length, were all measured at the ages of one.5 months, 6 months and 24 months. Appointment of nativity, birth weight and gender were obtained from midwife and hospital registers.
For the foetal ultrasound measurements, intra- and interobserver studies showed the intraclass correlation coefficient (ICC) to be higher than 0.98 and the corresponding coefficients of variation (CV) to be lower than 6%. Bland and Altman plots to test agreement of measurements demonstrated 95% limits of agreement to be within ten% departure from the mean of measurements, indicating good reproducibility [17]. For the postnatal ultrasound measurements, the intra-observer ICCs ranged from 0.93 (left and right renal widths and right renal thickness) to 0.99 (left renal length), and interobserver ICCs ranged from 0.64 (right renal thickness) to 0.90 (right renal length). Limits of agreement in the Banal and Altman plots ranged from −8.0% to 9.two% (intra-observer left renal width) to the widest limit from −eighteen.0% to xix.2% (interobserver left renal length) [18].
Data analysis
Differences in foetal and postnatal characteristics between boys and girls were assessed by t-tests and chi-foursquare tests for independent samples. Differences between left and right kidney were tested with paired-samples t-tests.
Data were analysed as recommended by Altman and Chitty [xix] and Royston and Altman [twenty]. For the reference curves for kidney growth, mail service-conceptional age was plotted against kidney length, width, depth and volume. From the original information, measurements of more than two standard deviations (SDs) from the regression line, fitted on our information, were considered to be outliers (north = 10) and were therefore removed. They were probably a result of measurement mistake or a data entry error. The best-plumbing equipment curves were determined by 2d-degree fractional polynomials [21]. We fitted the curves, using repeated measurement analyses and taking into account the dependency in the data by specifying a constant covariance between measurements of the same subject [twenty, 22]. The best-fitting fractional polynomial curves were chosen by our comparing the deviances, by Akaike's information criterion, and by our visually checking the goodness of fit. Next, regression lines were fitted for the dependency of the rest SD on conceptional historic period [23]. Subsequently, we plotted the SD scores confronting conceptional age to assess the correctness of the model.
Finally, centiles were derived and the curves were plotted on the data. The median age of two-year-onetime children visiting the research center was 25 months (95% range 23.six–28.3 months). Since only 34 children had undergone measurement beyond the postnatal historic period of 28 months (160 weeks afterward conception), the results are presented up to the postnatal age of 28 months. Reference curves for kidney growth were constructed for a post-conceptional age from xxx weeks to 160 weeks, corresponding to a gestational age of 30 weeks and a postnatal age of 28 months.
All statistical analyses were performed with the Statistical Package for the Social Sciences, version fifteen.0, for Windows (SPSS Inc, Chicago, IL, Us) and the Statistical Analysis System (SAS) for Windows, version 9.1.3.
Results
The percentage of boys was 52% (Table 2). The overall median age at the tertiary trimester of pregnancy was thirty weeks of gestation (total range 27.1–35.1 weeks). The overall median age of the infants at their 6-month postnatal visit was half dozen.3 months (total range 5.1–11.0 months), and at their 24-calendar month postnatal visit it was 25.1 months (total range 21.half dozen–31.6 months). Caput circumference at the third trimester and postnatal weight and length at the ages of 6 months and 24 months were larger in boys than in girls (all P values <0.001). No difference was found for gestational historic period at nascency betwixt boys and girls. In total, 15 children in our study group were born with a small size for gestational age [<−two standard difference scores (SDS)], 18 children had a depression birth weight (<2,500 g) and 23 children were born before term (gestational age <37 weeks). Table 3 shows that in the third trimester of pregnancy and at the age of 6 months all kidney measurements were larger in boys than in girls. At the age of 24 months, these gender differences were only meaning for left kidney structures and right kidney length. Both groups showed trends towards smaller left kidney measurements than right kidney measurements at all ages (Table iv).
Reference curves for individual measurements of kidney growth and fitted centiles are given in Fig. 1. Formulae for growth reference curves describing the mean with the corresponding standard difference are given in Tabular array five. Standard deviation increased linearly with gestational age. Reference values for kidney length, width, depth and volume are given in the Appendix (Tables 1S–4S).
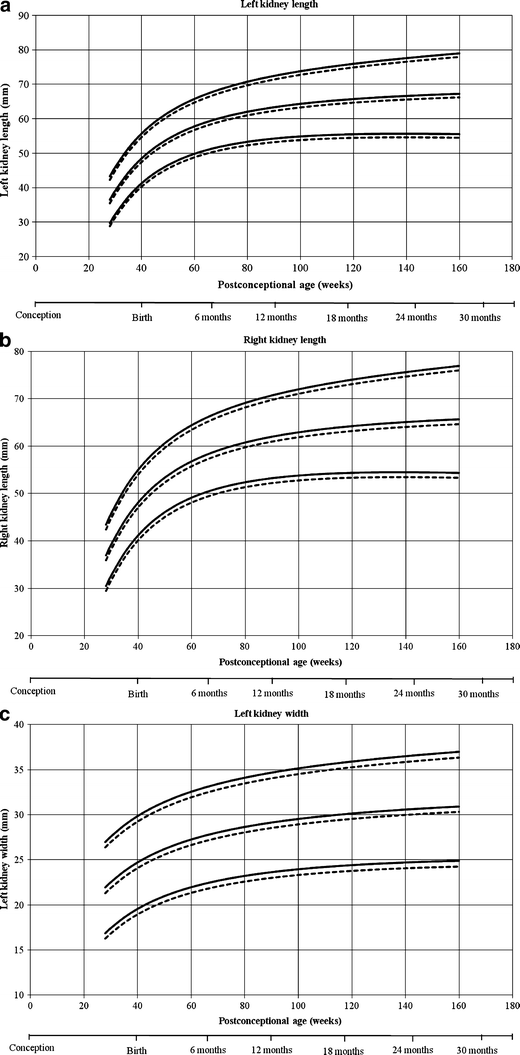
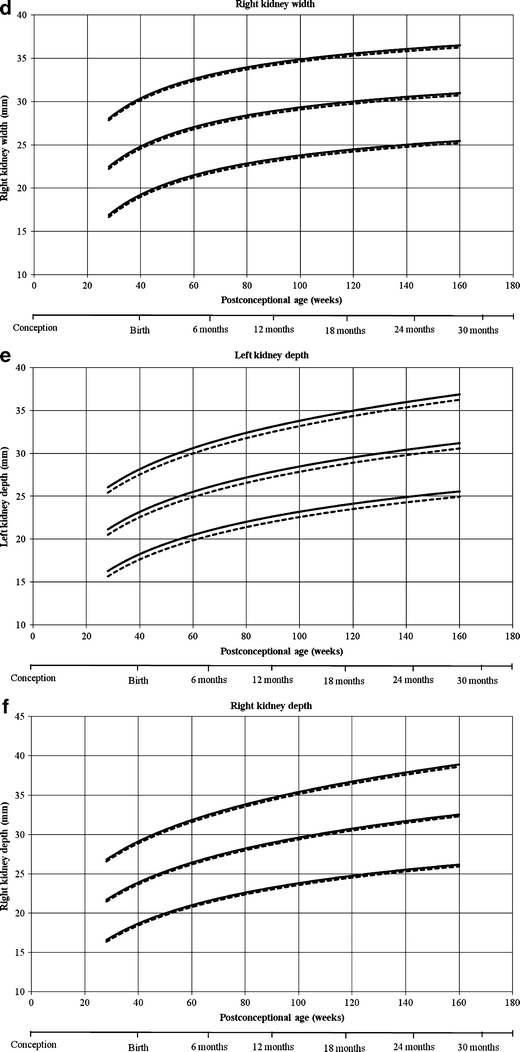
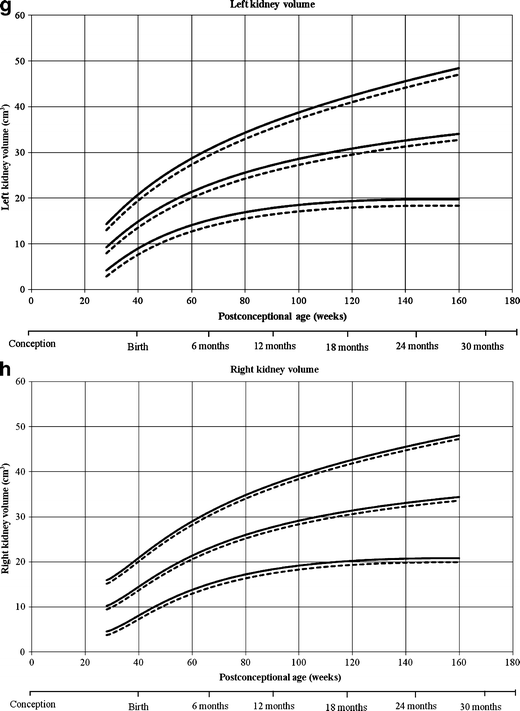
Reference growth curves of right and left kidney length, width, depth and volume in boys and girls, co-ordinate to mail-conceptional age. a Left kidney length, b correct kidney length, c left kidney width, d correct kidney width, e left kidney depth, f right kidney depth, k left kidney volume, h correct kidney book. Gender-differentiated growth curves for left and right kidney length, with, depth and volume measurements in relation to post-conceptional age with 3rd and 97th fitted centiles. The straight lines stand for boys and the dotted lines stand for girls
Word
We constructed gender-specific reference growth curves for kidney length, width, depth and book using measurements from a big population-based prospective accomplice study of healthy children followed from foetal life until early on childhood. We observed differences in kidney structures betwixt left and right kidneys and boys and girls.
The major forcefulness of our study is its prospective blueprint from foetal life and the size of the population-based cohort. Our reference curves were based on more two,500 kidney measurements. To our knowledge, no previous studies that focused on kidney size in early life were based on such large numbers. All foetal ultrasounds were carried out by two sonographers, and 86% of all postnatal ultrasounds were performed by one trained sonographer [24]. A limitation could be that in all children participating in the Generation R measurements at the ages of vi months and 24 months, kidney measurements were successfully performed in 83% and 80% of these infants, respectively. Missing values were mainly due to the infants crying or to the unavailability of equipment or radiographer. Our results would have been biased if the subjects' characteristics had differed between those included and those not included in the analyses. Nonetheless, we observed no differences in growth and foetal kidney characteristics between the subjects that had undergone postnatal kidney measurement and those that had not. Some other limitation could be that the written report was performed in a healthy population-based accomplice study. The pick towards a good for you population in our cohort might pb to a limited generalisability to preterm children or to children that were modest for gestational age at nascence. These numbers were also pocket-size to be assessed in farther particular.
In the tertiary trimester of pregnancy and at the age of 6 months all kidney measurements were larger in boys than in girls. At the age of 24 months, these gender differences were but pregnant for left kidney structures and right kidney length. Several studies of healthy neonates and adults have likewise shown that male subjects accept larger kidneys than those of female subjects [25, 26]. Ane explanation for this finding might exist a growth-stimulating issue of androgens, or Y-chromosome-related genes. Some other explanation could be that during foetal life testosterone levels are significantly higher in boys than in girls [27, 28].
Previously published information take shown alien results concerning differences betwixt left and right kidney size. Some studies found no divergence [29, 30], whereas others accept suggested the left kidney to be larger [31, 32]. Most consequent findings have been reported for kidney length, for which the left kidney seems to be longer than the right [7, 25, 26, 31, 33]. We constitute that the left kidney was longer in both boys and girls at the postnatal ages of half-dozen months and 24 months. In foetal life we establish no significant difference betwixt left and right kidney length.
Kidney growth is fastest during foetal life and early infancy, and the rate of increase gradually slows through the residue of the first year of life and finally stabilises [34]. In our report numbers and curves for the tertiary centiles showed decreasing kidney volume at older ages, from 140 weeks and onwards. The decreasing numbers are due to wider ranges, because of the low number of children with visits around 140 weeks and onwards. To deal with not-linear kidney growth, some sonographic standards, including means and standard deviations, for kidney size in relation to historic period have been published [34–36]. A few other studies created linear or non-linear polynomial regression equations for kidney size during the first year of life [29, 33]. One study created reference material for kidney size in salubrious children beyond the neonatal catamenia [7]. The authors only focused on kidney volume in relation to weight, height and body surface area and did not report data about prenatal kidney growth. To our noesis, our report is the offset to provide prospective longitudinal reference cloth for kidney size that covers the whole menses from foetal life to infancy in a healthy population.
In determination, kidney size differed between boys and girls from the age of 30 weeks of pregnancy until 24 months of age. The left kidney tended to be smaller than the right kidney. At the age of 24 months, the differences in right kidney size between boys and girls were attenuated. Gender-differentiated reference growth curves for both left and right kidneys were constructed for kidney length, width, depth and book. These reference curves may be of importance in the identification of abnormal kidney size and growth, with possible subsequent clinical consequences.
References
-
O'Neill WC (2000) Sonographic evaluation of renal failure. Am J Kidney Dis 35:1021–1038
-
Brenner BM, Chertow GM (1994) Congenital oligonephropathy and the etiology of adult hypertension and progressive renal injury. Am J Kidney Dis 23:171–175
-
Keller Chiliad, Zimmer G, Mall G, Ritz E, Amann K (2003) Nephron number in patients with principal hypertension. N Engl J Med 348:101–108
-
Geelhoed JJ, Verburg BO, Nauta J, Lequin M, Hofman A, Moll HA, Witteman JC, van der Heijden AJ, Steegers EA, Jaddoe VW (2009) Tracking and determinants of kidney size from fetal life until the historic period of ii years: the Generation R Study. Am J Kidney Dis 53:248–258
-
Gloor JM, Breckle RJ, Gehrking WC, Rosenquist RG, Mulholland TA, Bergstralh EJ, Ramin KD, Ogburn PL Jr (1997) Fetal renal growth evaluated past prenatal ultrasound examination. Mayo Clin Proc 72:124–129
-
Kennedy WA 2nd, Chitkara U, Abidari JM, Shortliffe LM (2003) Fetal renal growth as assessed through renal parenchymal area derived from prenatal and perinatal ultrasonography. J Urol 169:298–302
-
Schmidt IM, Main KM, Damgaard IN, Mau C, Haavisto AM, Chellakooty M, Boisen KA, Petersen JH, Scheike T, Olgaard K (2004) Kidney growth in 717 good for you children anile 0–18 months: a longitudinal cohort report. Pediatr Nephrol nineteen:992–1003
-
Spira EM, Jacobi C, Frankenschmidt A, Pohl M, von Schnakenburg C (2009) Sonographic longterm study: paediatric growth charts for single kidneys. Arch Dis Child 94:693–698
-
Pickworth FE, Carlin JB, Ditchfield MR, de Campo MP, de Campo JF, Cook DJ, Nolan T, Powell Hr, Sloane R, Grimwood K (1995) Sonographic measurement of renal enlargement in children with astute pyelonephritis and time needed for resolution: implications for renal growth assessment. AJR Am J Roentgenol 165:405–408
-
Parvex P, Willi JP, Kossovsky MP, Girardin E (2008) Longitudinal analyses of renal lesions due to acute pyelonephritis in children and their touch on on renal growth. J Urol 180:2602–2606
-
Jaddoe VW, Bakker R, van Duijn CM, van der Heijden AJ, Lindemans J, Mackenbach JP, Moll HA, Steegers EA, Tiemeier H, Uitterlinden AG, Verhulst FC, Hofman A (2007) The Generation R Report biobank: a resource for epidemiological studies in children and their parents. Eur J Epidemiol 22:917–923
-
Jaddoe VW, van Duijn CM, van der Heijden AJ, Mackenbach JP, Moll HA, Steegers EA, Tiemeier H, Uitterlinden AG, Verhulst FC, Hofman A (2008) The Generation R Written report: design and cohort update until the age of four years. Eur J Epidemiol 23:801–811
-
Verburg BO, Steegers EA, De Ridder M, Snijders RJ, Smith East, Hofman A, Moll HA, Jaddoe VWV, Witteman JCM (2008) New charts for ultrasound dating of pregnancy and assessment of fetal growth: longitudinal data from a population-based cohort report. Ultrasound Obstet Gynecol 31:388–396
-
Jeanty P, Dramaix-Wilmet Chiliad, Elkhazen N, Hubinont C, van Regemorter N (1982) Measurements of fetal kidney growth on ultrasound. Radiology 144:159–162
-
Gupta AK, Anand NK, Lamba IM (1993) Ultrasound evaluation of kidney dimensions in neonates. Indian Pediatr xxx:319–324
-
Konje JC, Okaro CI, Bell SC, de Chazal R, Taylor DJ (1997) A cross-sectional report of changes in fetal renal size with gestation in advisable- and small-for-gestational-age fetuses. Ultrasound Obstet Gynecol 10:22–26
-
Verburg BO, Mulder PG, Hofman A, Jaddoe VW, Witteman JC, Steegers EA (2008) Intra- and interobserver reproducibility study of early fetal growth parameters. Prenat Diagn 28:323–331
-
Geelhoed JJ, Kleyburg-Linkers VE, Snijders SP, Lequin Chiliad, Nauta J, Steegers EA, van der Heijden AJ, Jaddoe VW (2009) Reliability of renal ultrasound measurements in children. Pediatr Nephrol 24:1345–1353
-
Altman DG, Chitty LS (1997) New charts for ultrasound dating of pregnancy. Ultrasound Obstet Gynecol x:174–191
-
Royston P, Altman DG (1995) Pattern and analysis of longitudinal studies of fetal size. Ultrasound Obstet Gynecol 6:307–312
-
Royston P, Ambler M, Sauerbrei Westward (1999) The apply of fractional polynomials to model continuous take chances variables in epidemiology. Int J Epidemiol 28:964–974
-
Royston P (1995) Calculation of unconditional and conditional reference intervals for foetal size and growth from longitudinal measurements. Stat Med 14:1417–1436
-
Altman DG (1993) Structure of historic period-related reference centiles using accented residuals. Stat Med 12:917–924
-
Verburg BO, Geelhoed JJ, Steegers EA, Hofman A, Moll HA, Witteman JC, Jaddoe VW (2007) Fetal kidney book and its association with growth and blood flow in fetal life: The Generation R Study. Kidney Int 72:754–761
-
Emamian SA, Nielsen MB, Pedersen JF, Ytte Fifty (1993) Kidney dimensions at sonography: correlation with age, sex, and habitus in 665 developed volunteers. AJR Am J Roentgenol 160:83–86
-
Scott JE, Hunter EW, Lee RE, Matthews JN (1990) Ultrasound measurement of renal size in newborn infants. Arch Dis Child 65:361–364
-
Wood MG, de Peretti E, Lecoq A, Cadillon Due east, Zabot MT, Thoulon JM (1980) Concentration of 14 steroid hormones in human amniotic fluid of midpregnancy. J Clin Endocrinol Metab 51:816–822
-
Wilson JD, Griffin JE, Leshin Grand, George FW (1981) Role of gonadal hormones in evolution of the sexual phenotypes. Hum Genet 58:78–84
-
Han BK, Babcock DS (1985) Sonographic measurements and appearance of normal kidneys in children. AJR Am J Roentgenol 145:611–616
-
Holloway H, Jones TB, Robinson AE, Harpen Medico, Wiseman HJ (1983) Sonographic determination of renal volumes in normal neonates. Pediatr Radiol thirteen:212–214
-
Dinkel E, Ertel M, Dittrich Chiliad, Peters H, Berres G, Schulte-Wissermann H (1985) Kidney size in childhood. Sonographical growth charts for kidney length and volume. Pediatr Radiol 15:38–43
-
Schmidt IM, Molgaard C, Principal KM, Michaelsen KF (2001) Outcome of gender and lean body mass on kidney size in good for you 10-year-old children. Pediatr Nephrol 16:366–370
-
Konus OL, Ozdemir A, Akkaya A, Erbas G, Celik H, Isik South (1998) Normal liver, spleen, and kidney dimensions in neonates, infants, and children: evaluation with sonography. AJR Am J Roentgenol 171:1693–1698
-
Vujic A, Kosutic J, Bogdanovic R, Prijic Due south, Milicic B, Igrutinovic Z (2007) Sonographic assessment of normal kidney dimensions in the offset year of life—a study of 992 healthy infants. Pediatr Nephrol 22:1143–1150
-
Rosenbaum DM, Korngold E, Teele RL (1984) Sonographic cess of renal length in normal children. AJR Am J Roentgenol 142:467–469
-
Zerin JM, Meyer RD (2000) Sonographic assessment of renal length in the first year of life: the problem of "spurious nephromegaly". Pediatr Radiol 30:52–57
Acknowledgements
The Generation R Report was conducted by the Erasmus Medical Middle in close collaboration with the School of Law and the Faculty of Social Sciences at the Erasmus Academy, Rotterdam, the Municipal Wellness Service, Rotterdam surface area, the Rotterdam Homecare Foundation, and the Stichting Trombosedienst & Artsenlaboratorium Rijnmond (STAR), Rotterdam. Nosotros gratefully admit the contribution of general practitioners, hospitals, midwives and pharmacies in Rotterdam.
The starting time phase of the Generation R Study was made possible by financial support from the Erasmus Medical Eye, Rotterdam, the Erasmus Academy, Rotterdam, and the Netherlands Arrangement for Wellness Research and Development (ZonMw). Boosted support was provided by a grant from the Dutch Kidney Foundation (C08.2251).
The authors declare that they have no personal or financial conflicts of interest.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits whatever noncommercial utilize, distribution, and reproduction in whatever medium, provided the original author(s) and source are credited.
Author information
Affiliations
Corresponding writer
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This is an open access commodity distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/ii.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Reprints and Permissions
Well-nigh this commodity
Cite this commodity
Geelhoed, J.J.M., Taal, H.R., Steegers, E.A.P. et al. Kidney growth curves in salubrious children from the third trimester of pregnancy until the age of two years. The Generation R Study. Pediatr Nephrol 25, 289–298 (2010). https://doi.org/ten.1007/s00467-009-1335-2
-
Received:
-
Revised:
-
Accustomed:
-
Published:
-
Issue Date:
-
DOI : https://doi.org/10.1007/s00467-009-1335-ii
Keywords
- Kidney dimensions
- Reference growth curves
- Children
- Ultrasound
- Gender
Source: https://link.springer.com/article/10.1007/s00467-009-1335-2
0 Response to "My Baby Has Lower Left Kidney Than Right"
Post a Comment